Horrifying medical device malfunction: Abdominal implant erupts from leg
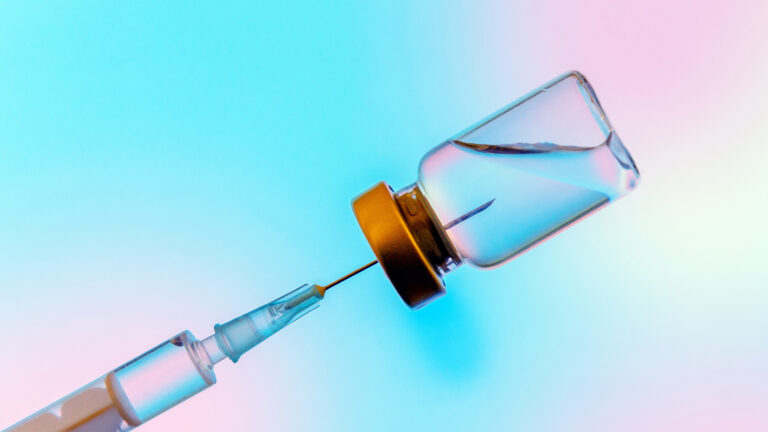
On May 7, 2011, Georgia resident Tonya Brand noticed a pain on the inside of her right thigh. As the pain grew worse in the 4- to 5-inch area of her leg, she headed to a hospital. There, doctors suspected she had a blood clot. But an ultrasound the next day failed to find one. Instead, it revealed a mysterious toothpick-sized object lodged in Brand's leg.
Over the next few weeks, the painful area became a bulge, and on June 17, Brand put pressure on it. Unexpectedly, the protrusion popped, and a 1.5-inch metal wire came poking out of her leg, piercing her skin.
The piece of metal was later determined to be part of a metal filter she had implanted in a vein in her abdomen more than two years earlier, in March 2009, according to a lawsuit Brand filed. The filter was initially placed in her inferior vena cava (IVC), the body's largest vein tasked with bringing deoxygenated blood from the lower body back up to the heart. The filter is intended to catch blood clots, preventing them from getting into the lungs, where they could cause a life-threatening pulmonary embolism. Brand got the IVC filter ahead of a spinal surgery she had in 2009, which could boost her risk of clots.
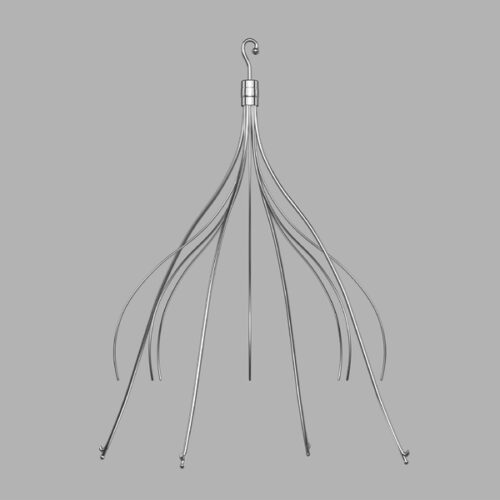
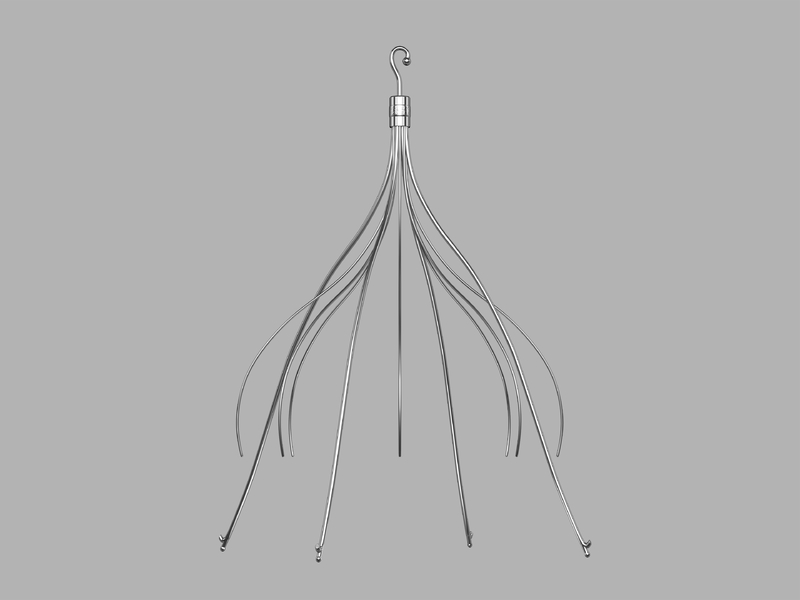
© Cook Medical